|
10/28/2022 0 Comments Reverse phase hplc Failure to remove the buffer salt will cause it to precipitate out of solution at the next step, which will result in a pressure increase and clogging of column frits.Įven after proper equilibration, many analysts still notice more interference or inaccuracy with the first injection after a system has been sitting idle. (If using an Aqueous C18 phase, you may use up to 100% aqueous if needed). However, a word of caution: If you are using buffer salts, make sure to flush them out with at least 50% but no higher than 90% aqueous solution before gradually increasing the organic content. This usually minimizes those effects if they occur. It is also a good practice to use if you are noticing an elevated baseline by UV that you suspect might be column bleed. Pumping a slightly higher organic content as described above is also a good practice to use for columns that have sat idle for a while, especially those that have had dirty sample matrices loaded. After you do this, you can gradually begin pumping your mobile phase to equilibrate. This is a precaution in case there is anything present that has a higher solubility in the new solvent versus the previous one(s). If you are using a gradient, using a mobile phase that is higher in organic content than your storage solvent, or if you are using a mobile phase that has a different organic solvent than what it was stored in (for example, acetonitrile instead of methanol), you should pump for a while with a solution that contains the organic solvent you will be using in a slightly higher (10-20% higher) content than what your method calls for. How do I determine total column volume or void volume for LC? If you need help determining column volume, click on the link below to go through the calculations: If you will be doing isocratic runs and your mobile phase is similar to the solvent used for storage and shipping (usually 50/50 methanol and water), you should only need to pump 7 -10 column volumes of mobile phase through the column. You do, however, need to equilibrate in the mobile phase that you will be using. There is no baking at high temperatures involved like you might do for GC or that sort of thing. Well, it’s partly a matter of semantics sometimes, but there is usually nothing drastic that you need to do to get your HPLC column going if it is new. ASE Extraction System Parts Cross-Reference.Documentation Search (SDS, Certs, Data Packs).EZGC Method Translator & Flow Calculator.A large number of biological molecules fall into this category such as amino acids, peptides, proteins, nucleic acid and oligosaccharides.Īdvantages of Liquid Chromatography (LC) vs. 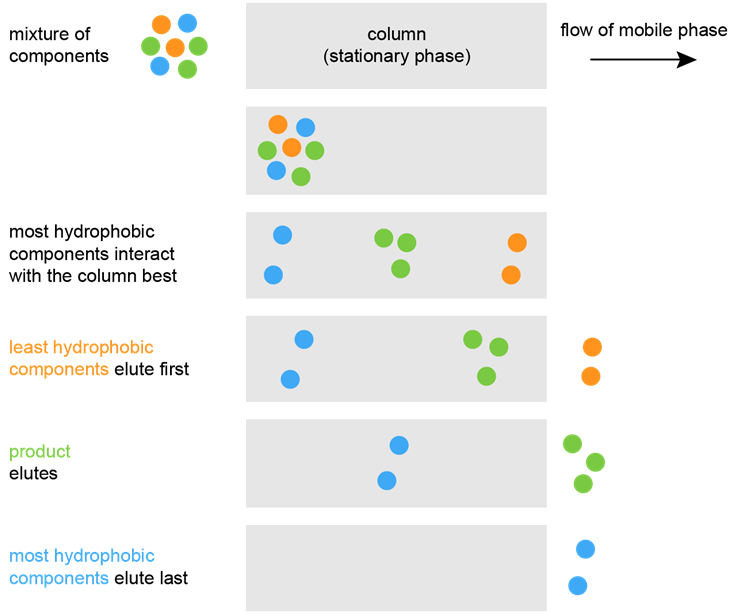
The molecule releases from the stationary phase and elutes.īecause of the fact that the mobile phase in reverse phase chromatography is polar, reverse phase chromatography is applied to the separation and determination of polar molecules that either are insoluble in organic solvents or are attached too strongly to the polar stationary phase in normal phase chromatography. A buffer of increasing hydrophobicity is used to dissociate the bound molecule at a point at which the hydrophobic interaction between the exposed groups and the stationary phase is less favorable than the interaction between the bound molecule and the solvent. This partitioning occurs as a result of the solute molecule tending to have hydrophobic groups, and binding via those groups to the hydrophobic stationary phase. I.1 and has to do with the binding of a solute - having both hydrophobic and hydrophilic parts (groups) - to a stationary hydrophobic molecule (stationary phase) in a polar solvent. The mechanism in reverse phase separation is shown in Fig. This chromatographic technique is far superior to the other modes of separation in the variety of target compounds it can analyze. The name “ reversed phase” is derived from the opposite technique of “ normal phase” chromatography which involves the separation of molecules based upon their interaction with a polar matrix (silica beads) in the presence of a nonpolar mobile phase (nonpolar solvent). In reverse phase the stationary phase is nonpolar and the mobile phase is polar. It is used to separate nonpolar molecules in solution. Reverse phase chromatography is the most commonly used LC or HPLC separation mode. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
